 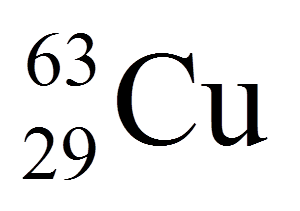 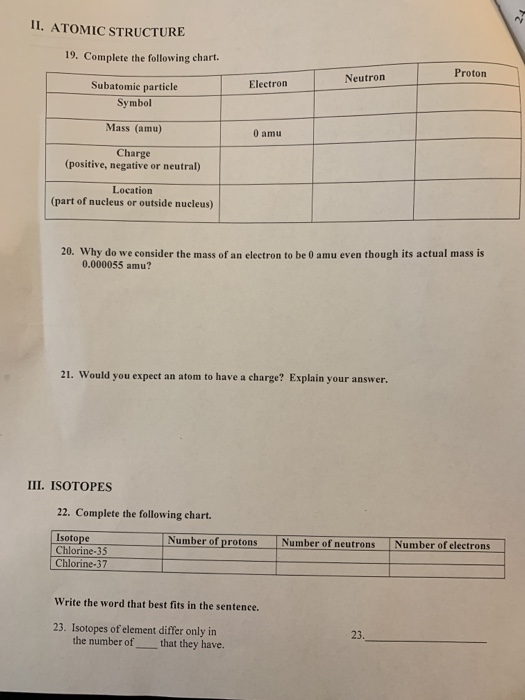
Pejman Rowshanfarzad Mahsheed Sabet AmirReza Jalilian Mohsen Kamalidehghan (2006).Application of Copper radioisotopes in Medicine (Review Paper):.CRC Handbook of Chemistry and Physics (85th ed.). Half-life, spin, and isomer data selected from the following sources.International Union of Pure and Applied Chemistry. "News & Notices: Standard Atomic Weights Revised"."Atomic weights of the elements 2005 (IUPAC Technical Report)". de Laeter, John Robert Böhlke, John Karl De Bièvre, Paul Hidaka, Hiroshi Peiser, H.Isotopic compositions and standard atomic masses from:.Audi, Georges Bersillon, Olivier Blachot, Jean Wapstra, Aaldert Hendrik (2003), "The N UBASE evaluation of nuclear and decay properties", Nuclear Physics A, 729: 3–128, Bibcode: 2003NuPhA.729.3A, doi: 10.1016/j.nuclphysa.2003.11.001."Clarity uses a cutting-edge imaging technique to guide drug development". ^ a b Audi, Georges Bersillon, Olivier Blachot, Jean Wapstra, Aaldert Hendrik (2003), "The N UBASE evaluation of nuclear and decay properties", Nuclear Physics A, 729: 3–128, Bibcode: 2003NuPhA.729.3A, doi: 10.1016/j.nuclphysa.2003.11.001."Standard atomic weights of the elements 2021 (IUPAC Technical Report)". ^ Prohaska, Thomas Irrgeher, Johanna Benefield, Jacqueline et al."The NUBASE2020 evaluation of nuclear properties" (PDF). There is a growing interest in the use of 64Cu, 62Cu, 61Cu, and 60Cu for diagnostic purposes and 67Cu and 64Cu for targeted radiotherapy.įor example, 64Cu has a longer half-life than most positron-emitters (12.7 hours) and is thus ideal for diagnostic PET imaging of biological molecules. ^ a b # – Values marked # are not purely derived from experimental data, but at least partly from trends of neighboring nuclides (TNN).Ĭopper offers a relatively large number of radioisotopes that are potentially suitable for use in nuclear medicine.^ ( ) spin value – Indicates spin with weak assignment arguments. 
^ Bold symbol as daughter – Daughter product is stable.^ # – Atomic mass marked #: value and uncertainty derived not from purely experimental data, but at least partly from trends from the Mass Surface (TMS).^ ( ) – Uncertainty (1 σ) is given in concise form in parentheses after the corresponding last digits.The least stable is 69mCu with a half-life of 360 ns. The most stable of these is 68mCu with a half-life of 3.75 minutes. 70Cu has two isomers, making a total of 7 distinct isomers. Ħ8Cu, 69Cu, 71Cu, 72Cu, and 76Cu each have one metastable isomer. Unstable copper isotopes with atomic masses below 63 tend to undergo β + decay, while isotopes with atomic masses above 65 tend to undergo β − decay. The most stable radioisotope is 67Cu with a half-life of 61.83 hours, while the least stable is 54Cu with a half-life of approximately 75 ns. Copper ( 29Cu) has two stable isotopes, 63Cu and 65Cu, along with 27 radioisotopes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
